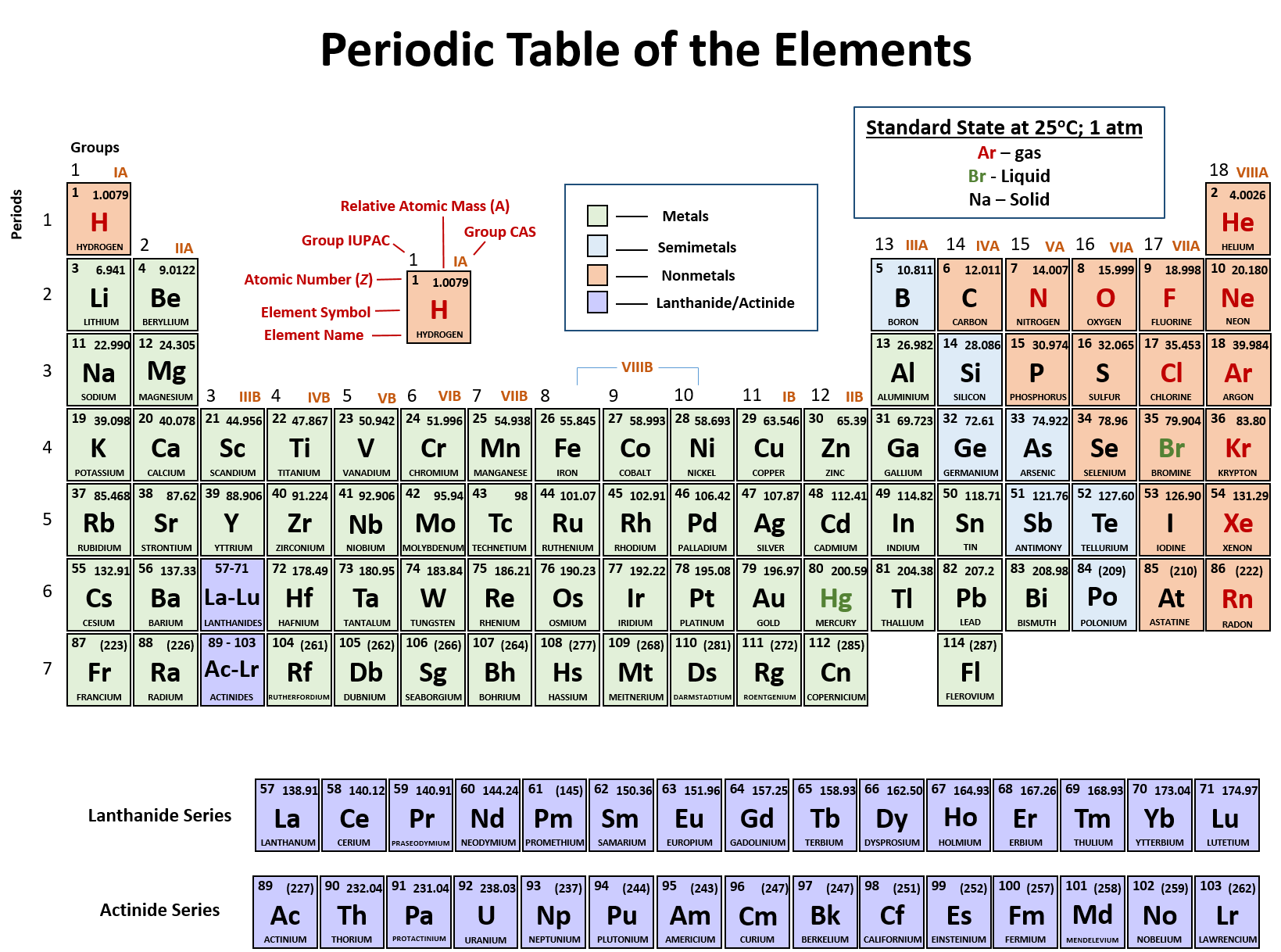
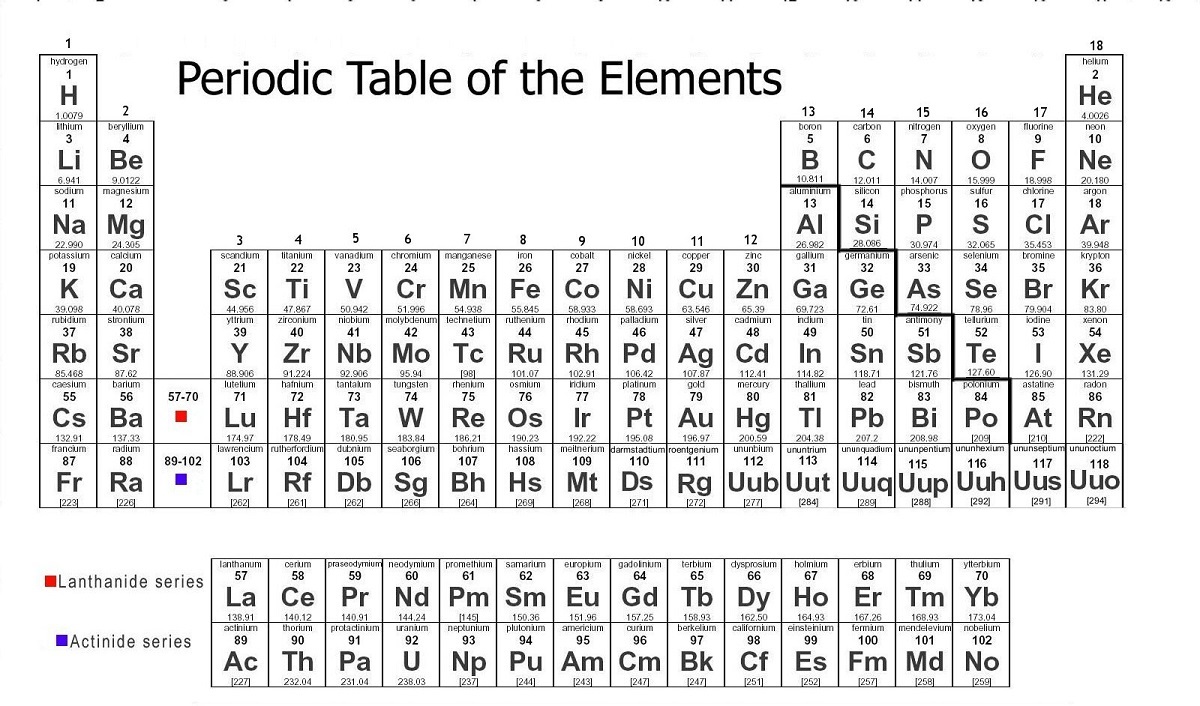
Example: Carbon-12įor example, the most common form (isotope) of carbon-12 is written as: So, if I wrote just the element symbol and its atomic mass on the board that students should be able to figure out the number of particles. And to draw the atoms you fill up the inner shells first then move on to the outer shells. Each shell can hold a certain maximum number of electrons (2 for the first shell 8 for the second shell and 8 for the third). This oxygen atom has 8 electrons in two shells.Įlectron Shells: Electrons orbit around the nucleus in a series of shells. 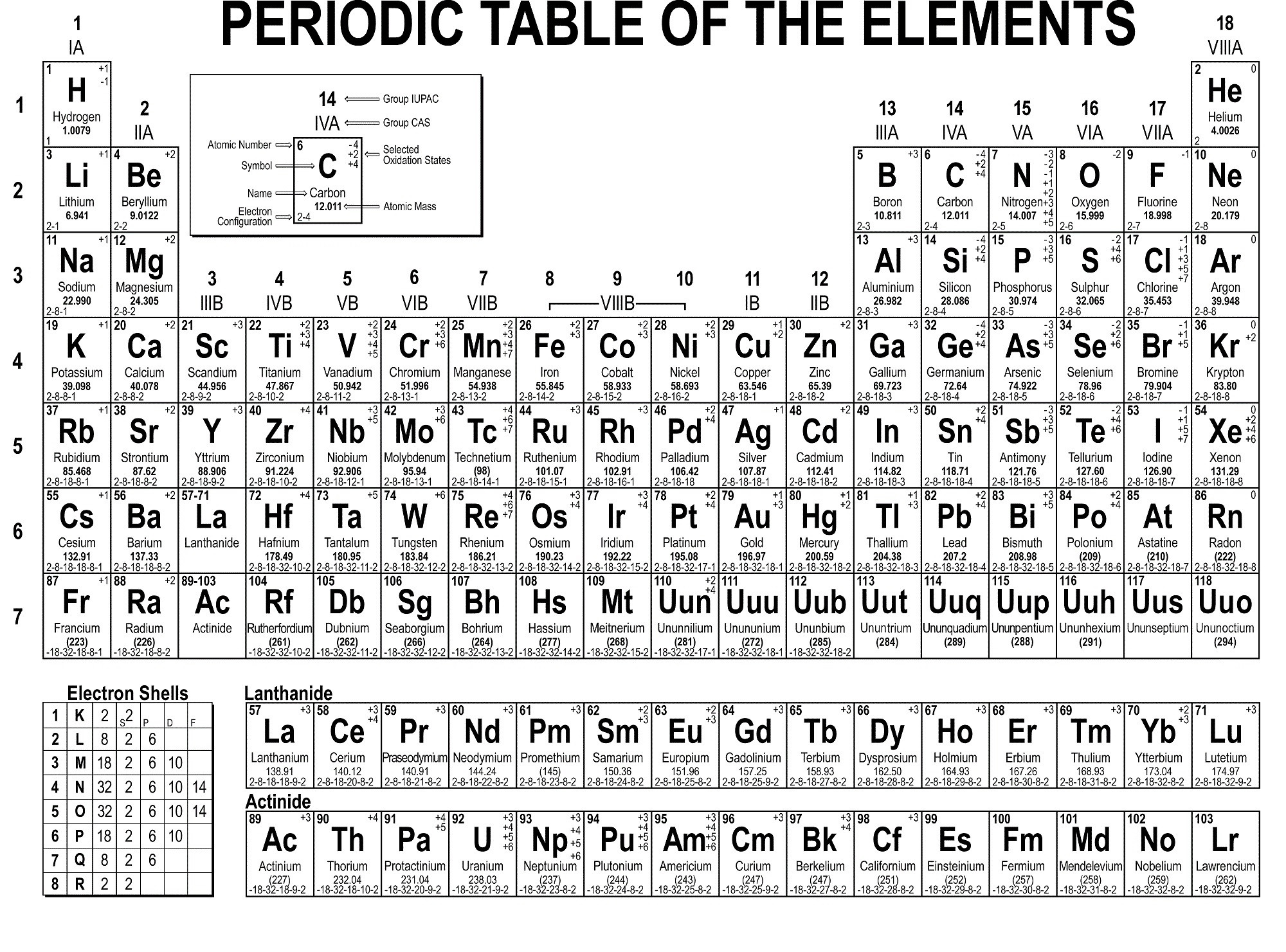
(Note that electrons don’t contribute to the mass of the atom because their masses are so much smaller than the masses of neutrons and protons. 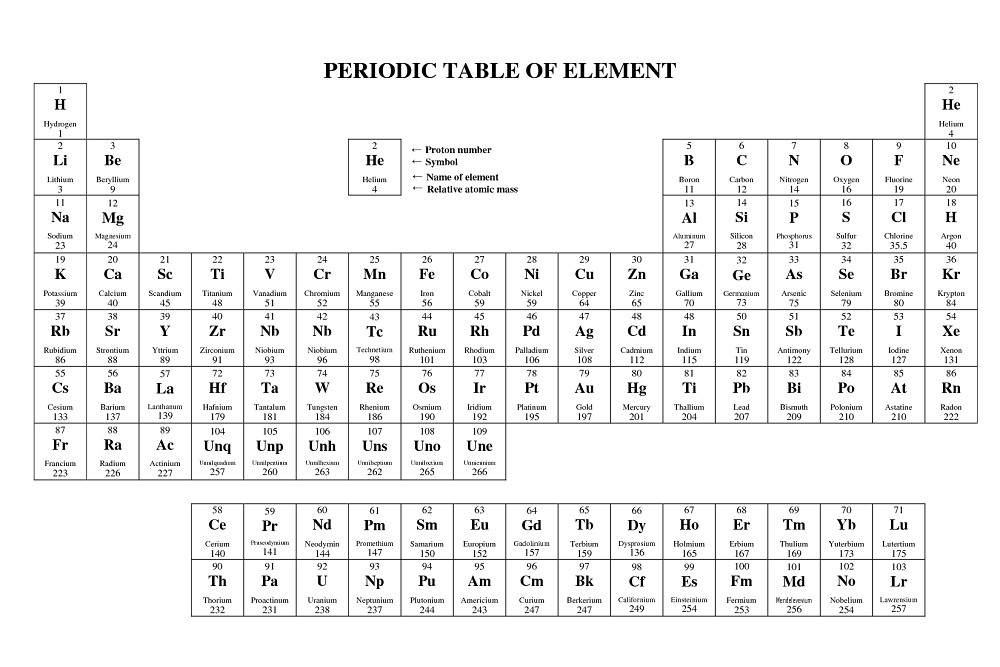
Since the atomic mass is the sum of the number of protons and neutrons, if you know the atomic mass and the number of protons, you can easily figure out the number of neutrons. So how do you know how many neutrons? You have to ask, or look at the atomic mass number, which is usually written to the upper left of the atom. The small atoms that we’re looking at tend to have the same number of neutrons as protons, but that’s not necessarily the case. The atomic mass is the sum of the number of protons (2) and the number of neutrons (2).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
